|
Microbes often produce not just one member of a metabolite class but a complex mixture of analogues, metabolites with closely related
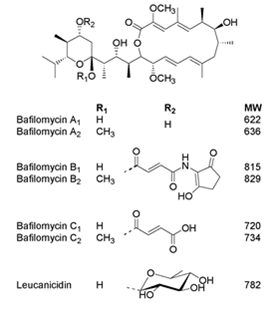
chemical structures. The bafilomycins provide an interesting example. A typical bafilomycin producing Streptomyces will yield a range of analogues: bafilomycins
A1, B1, C1 and D, with A1, B1 and C1 able to be converted non-enzymatically to A2, B2 and C2. Why does the microorganism expend the
energy to produce so many closely related chemicals? Why make metabolites which are readily modified by non-enzymatic means to provide
further structural diversity? Does this production of multiple analogues reflect a redundancy in nature? Or does the presence of so many
analogues serve a purpose? |
|
Bafilomycins are active against bacteria, fungi, insects, nematodes and mammalian cells, but
the potency of these activities varies from analogue
to analogue, with each having a distinct pattern of selectivity for these targets. Microbes
have learnt that there can be subtle variations in the
structures of the receptors with which their metabolites interact. By providing a range of
analogues bafilomycin producers appear to be ensuring
a broad inhibition across a range of variations or 'sub-types' in the bafilomycin receptor site,
the significance of which we are yet to grasp.
Exploring structure-activity relationships, the way in which a biological activity varies with
subtle differences in the structure of an inhibitor, provides the most readily accessible route
to understanding micro-diversity within receptor function. Microbes often provide us with a
ready supply of related structures but most of the analogues reported are largely ignored,
while research focuses on just a single analogue.
The availability of analogues of known actives offers considerable potential to help
characterise selectivity within receptor sub-types.
Yet, until now, access to many of these metabolites has been non-existent.
|
|